April 11, 2023 Ι The highly toxic heavy metal thallium (Tl) occurs as a trace element in the terrestrial environment with an average concentration of 490 µg/kg. It is found in particular as an isomorphous admixture in sulfidic ores.
Through the mining of such ores and subsequent industrial treatment steps (e.g. smelting), Tl enters the environment, also non-anthropogenically through leaching of sedimentary rocks [1], [2]. Aquatic Tl contamination is an environmental problem, especially in emerging countries. Here, in rivers close to opencast mining areas, sulphide ores have Tl contents of > 1000 µg/L [3]. In contrast, the guidelines for the EU environmental quality standards for surface water provide for a Tl-limit value of 0.2 µg/ L [4].
Combining water purification with resource recovery
A research goal within the INNO-KOM project “Thallium oxide production / thallium removal from water” funded by the German Federal Ministry of Economics and Climate Protection is the development of a practical and economically feasible process for the immobilisation of Tl contamination from water.
The adsorption strategy implemented by the IUTA [5] for this purpose consists in the synthesis of a binary sorbent system, which initially involves an intrinsic oxidation of the Tl+ to efficiently immobilize Tl+++ causes before sorption occurs. The sorbent system produced consists of inexpensive iron (Fe) and manganese (Mn) compounds that can be easily reacted and whose reaction product is in line with the treatment substances permitted under the German Drinking Water Ordinance.
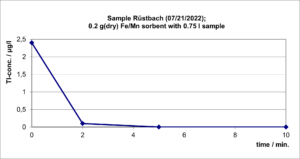
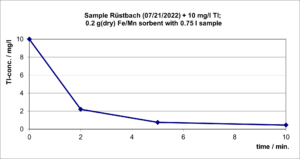
This Fe/Mn sorbent has been tested in batch experiments with a water sample taken from the Rüstbach (Stolberg), with the original sample (2.4 µg/L Tl) and an identical but around 10 mg/L Tl – spiked Rüstbach sample were used. The experiments were carried out with magnetic stirring with 0.2 g (dry) Sorbent and 750 ml water phase for 10 minutes each. Sampling found after 0; 2; 5 and 10 minutes with immediate filtration.
For the original Rüstbach sample, the tests showed a Tl reduction below the analytical limit of quantification (0.05 µg/l). The sample spiked 4000 times with the natural Tl content could be reduced by 95% of the Tl. Figures 1 and 2 show the decay of the Tl content over time.
The results show that traces of Tl in water can be safely removed by adsorption using the easily available Fe / Mn sorbent. In the next trials, a hybrid process consisting of sulfide precipitation with subsequent adsorptive Tl and excess sulfide fine cleaning will be tested, which can be used for the internal treatment of waste water containing heavy metals.
Short publication by Dipl.-Chem. Dipl.-Ing. (FH) Frank Grüning
IUTA Institut für Energie und Umwelttechnik e. V.
You will find the German-language article in the next issue of gwf Wasser/Abwasser 04/2023.
Literature:
- Zhuang W, Song J Thallium in aquatic environments and the factors controlling Tl behavior; Environ Sci Pollut Res 28, 35472-35487 (2021)
- Nriagu , JO (Editor): Thallium in the environment, John Wiley & Sons, Inc., New York 1998
- Belzile , N., Chen, Y.-W.: Thallium in the environment: A critical review focused on natural waters, soils, sediments and airborne particles; Applied Geochemistry, 84, 218-243 (2017)
- Available at: https://www.umweltbundesamt.de/themen/wasser/fluesse/status/metalle
- Institut für Energie- und Umwelttechnik e. V., Bliersheimer Str. 58-60, 47229 Duisburg