Polyfluorinated chemicals exist in numerous molecular variations, their applications range from non-stick pans over fire extinguishing foams and food packaging to water repellent textiles, etc. They can resist high temperatures, corrosive products and electricity, and they are water and dirt-repellent. However, due to this high molecular stability, these chemicals are biologically non-degradable, that is why they are referred to as ‘forever-chemicals’. Fluorine pollution is a wide spread issue; the chemicals disperse via the air and water, ending up in our food and drinking water. There is also evidence that PFAS can damage our health in the long term, as the substances are potentially carcinogenic and hormonally disruptive.
Selective filter for very low concentrations
The doctoral research of Matthias Van den Bergh, under the supervision of Professor Dirk De Vos and in collaboration with VITO, may provide a solution in the long term. “The big challenge is that these PFAS molecules are only present in the water in very low concentrations”, explains Professor De Vos. “In the case of heavy contaminations we’re talking about an order of magnitude of one microgram per litre. So it’s like trying to find a needle in a haystack.”
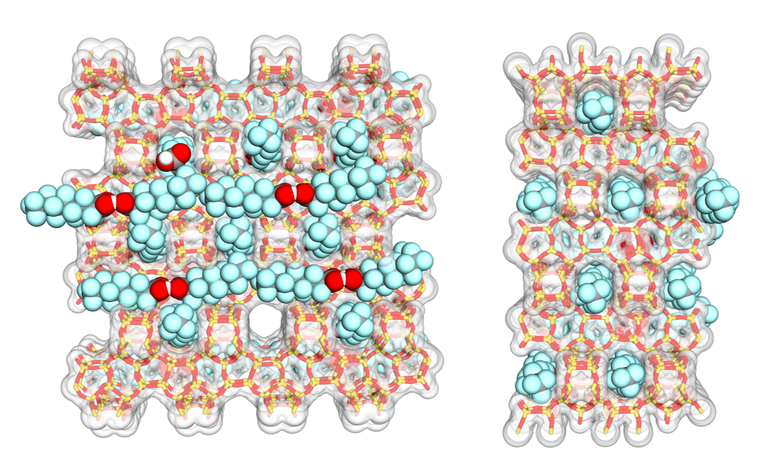
“Nevertheless, we have succeeded in developing an efficient filter material”, says Matthias Van den Bergh. “We’re using a zeolite, a porous substance with tiny cavities. This material, in powdered form, attracts the PFAS molecules and traps them in its pores. The great thing about this technology is that it works very selectively: the harmful PFAS substances are filtered, but the other harmful components in the water are not.”
Patented concept
The researchers have patented the concept, but there is still a long way to go to achieve a working product, explains Professor Dirk De Vos. “We strongly believe in the possibilities of this material, but the challenge is to produce it on a large scale at a reasonable cost. We’re also going to further investigate in what way and in what form the filter is best used. So there’s no ready-made solution yet, but it is important that steps are taken to address this underexposed form of pollution.”
The research work was published in Angewandte Chemie. It is available under https://doi.org/10.1002/anie.202002953